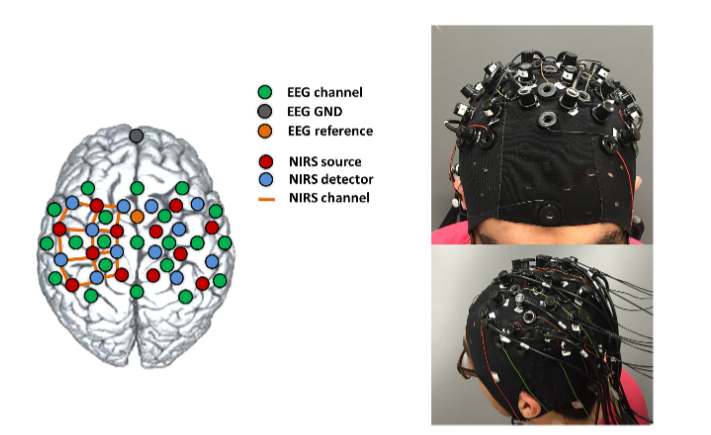
Concurrent fNIRS and EEG
The NIRSport2 combined with the g.Nautilus PRO FLEXIBLE from g.tec
The NIRSport2 combined with the LiveAmp from Brain Products.
Motivation
A local increase in cortical blood flow accompanies almost all neuronal responses to stimuli in the brain. This relationship, termed neurovascular coupling, involves many steps from the initial firing of the neurons, to release of chemical transmitters, to the final vasoconstriction or vasodilatation. Understanding this relationship between brain activity and the resulting changes in metabolism and blood flow remains a vital research area.
EEG and fNIRS capture different events in this cascade but are both linked to the same neural activity. Thus, the combination of these two methodologies offers the possibility to examine the complex patterns of cortical activity more comprehensively. Furthermore, each modality can serve as a built-in validation for activity identified in the other.
Meanwhile, EEG and fNIRS have very different but complementary temporal and spatial resolution: While Evoked Potentials (EPs) detect the cortical response to a given stimulus with high temporal resolution, fNIRS localizes changes in oxygen metabolism that follow neural activation. Thus, when recorded simultaneously, you can leverage the strengths of both technologies.
Sensor placement & EEG system compatibility
Integration with Brain Products EEG system
Integration with g.tec EEG system
Combining EEG and fNIRS is not only scientifically powerful, but also straightforward to implement with NIRx fNIRS systems. Thanks to the optical nature of the fNIRS signal, the compact active optode design, and advanced cable routing procedures, fNIRS can be integrated with EEG without mutual interference or complex technical adjustments.
NIRx fNIRS systems are designed to integrate seamlessly with most research-grade EEG setups. Integration is achieved simply by placing EEG electrodes and fNIRS optodes next to each other on the same cap, without the need for specialized adapters or custom hardware. We use EEG caps manufactured by EASYCAP to position our optodes, allowing researchers to continue working withtheir preferred EEG electrodes and amplifiers while keeping setup time short and workflows familiar.
The cap montage is fully customizable, making it easy to adapt the sensor layout to different research questions, paradigms, and participant groups. For optimal data quality, we recommend combining NIRx fNIRS optodes with gel-based active or actively shielded EEG electrodes, but the system remains flexible enough to accommodate a wide range of EEG configurations.
Mobile fNIRS/EEG setup with NIRSport2 & TMSi SAGA system using actively shielded electrodes
If you are wondering whether your EEG system is compatible with our fNIRS systems and how to achieve integration, please don’t hesitate to reach out to us.
Data Synchronization
As fNIRS and EEG measurements are recorded independently, it is important to ensure simultaneous trigger acquisition with both data streams. This precisely marks the data segments of interest for online and offline analysis.
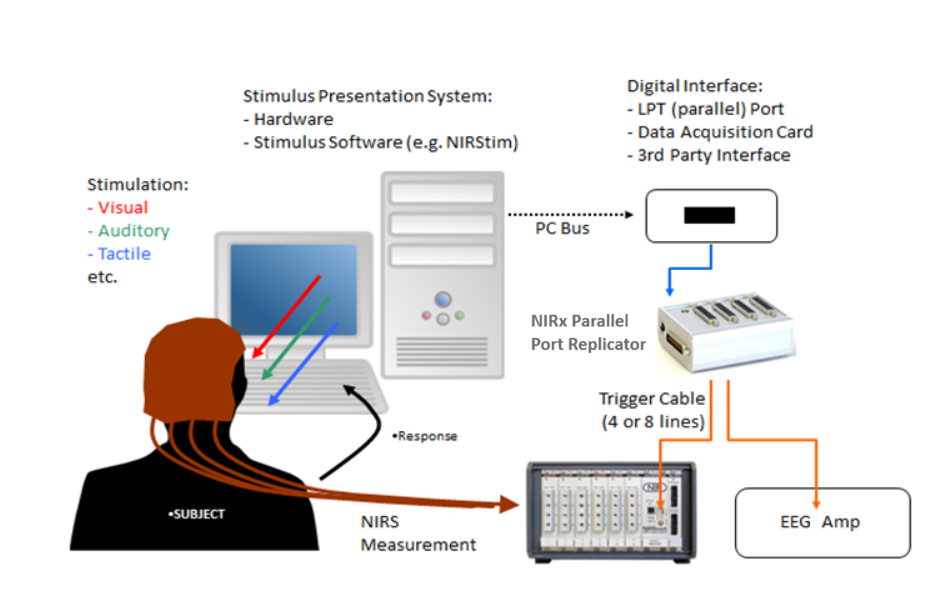
A precise synchronization using trigger cables can be easily achieved with the NIRx Parallel Port Replicator [more details here], which powers the incoming signal via USB and splits a single DB-25 (parallel port) input to four or more outputs.
A general schematic of stimulus presentation and synchronization of fNIRS and EEG measures is depicted to the right below.
Above: Example set up of EEG/fNIRS trigger synchronization using the NIRx Parallel Port Replicator.
If you prefer a fully wireless and mobile setup, you can send synchronized event markers to both, the EEG and the fNIRS system at the same time using a wireless protocol called Lab Streaming Layer (LSL) [more details here]. All our devices accept LSL trigger streams, so you only need to ensure that your EEG device does as well.
Application examples
Below you can find two selected examples of how fNIRS and EEG can be applied in a combined setup to leverage the strengths of both technologies.
1. fNIRS-informed EEG Source Imaging (Cao et al., 2023)
In this study a 45-degree-wide black-and-white checkerboard wedge was used as the stimulus, which was displayed alternatingly on the left- and right-hand visual field (Stim A and Stim B) to elicit activation in the contralateral side of the visual cortex. Meanwhile, 64-channel EEG and HD-DOT were recorded simultaneously over occipital and parietal areas (for more information about HD-DOT see our blog post on HD-fNIRS):
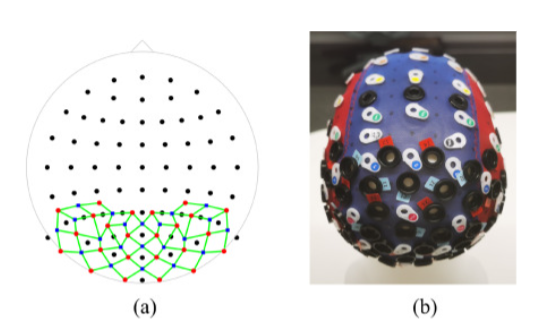
Description: “Fig. 2. The configuration of DOT and EEG used in the experiments. (a) A 2D illustration of the setup. Black dots: EEG electrodes; red dots: DOT sources; blue dots: DOT detectors; green lines, DOT channels. (b) The cap used in the experiments mounted on a mannequin. White rings: BioSemi EEG electrode holders; black rings in the back: NIRx optode holders; black rings in the middle: locations where the accelerometer and the cable organizers were installed.”. Reproduced from Cao et al., 2023.
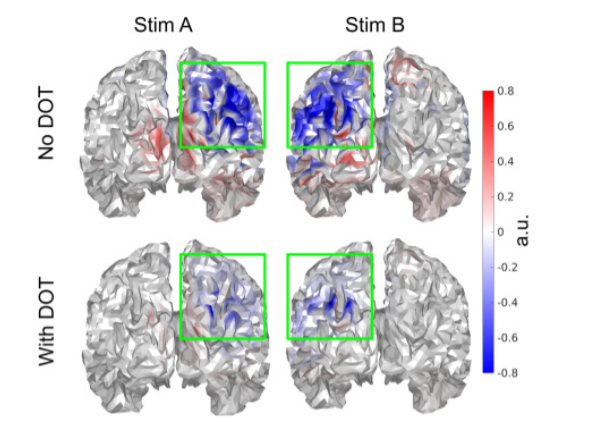
The results suggest that using the activation as measured by HD-DOT as a spatial prior in the EEG source reconstruction improved the spatial accuracy:
Description: “Fig. 4. Improved spatial confinement of source reconstruction at the respective N170 peaks when using DOT as the spatial prior. Each subfigure was individually normalized between -1 and 1 for better clarity. Color bars slightly saturated for better visualization. Only voxels with p<0.05 are displayed. The standard Colin27-based brain surface from Fieldtrip is used.”. Reproduced from Cao et al., 2023.
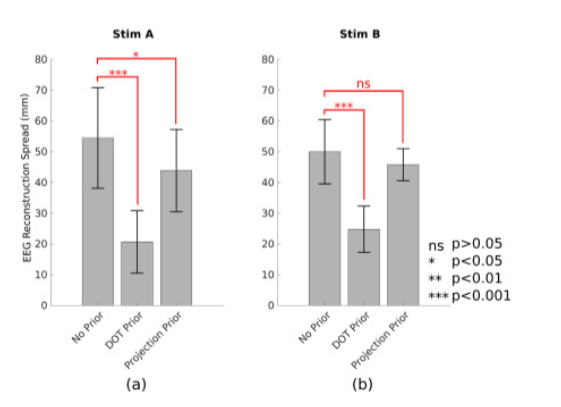
In addition to using HD-DOT and no priors at all in the EEG source reconstruction, the authors also used only ∆HbO as a projection prior to investigate whether there is a reduction of the EEG reconstruction spread. Here is what they found:
Description: “Fig. 8. Comparison of group level spread metric using no prior, DOT prior, and HbO projection prior. Shown are group averages, standard deviations, as well as results of statistical testing for the two stimuli separately.” Reproduced from Cao et al., 2023.
This study highlights the potential of the two modalities to complement each other: HD-DOT/fNIRS alone could not have measured the cortical activation induced by the stimulus due to the fast frequency at which the stimulus presented, while employing EEG source imaging without the spatial priors overestimates the spread of the activation. Combining the two modalities thus combines the high spatial resolution of fNIRS and HD-DOT with the excellent temporal resolution of the EEG, leading to improved results.
2. fNIRS–EEG Feature Fusion for Classification in a BCI (Buccino et al., 2016)
In this study the authors tested the classification accuracy of a BCI using EEG and fNIRS separately, as well as a combination of features from both modalities to distinguish four different executed movements: Right-Arm, Left-Arm, Right-Hand and Left-Hand. They used a probe setup with EEG electrodes and fNIRS optodes located bilaterally over the motor cortices:
Description: “Fig 1. Left: EEG electrodes and fNIRS optodes configuration on the cap. Right: Real picture of a subject wearing the cap completely mounted (with EEG electrodes, fNIRS sources and detectors).” Reproduced from Buccino et al., 2016.
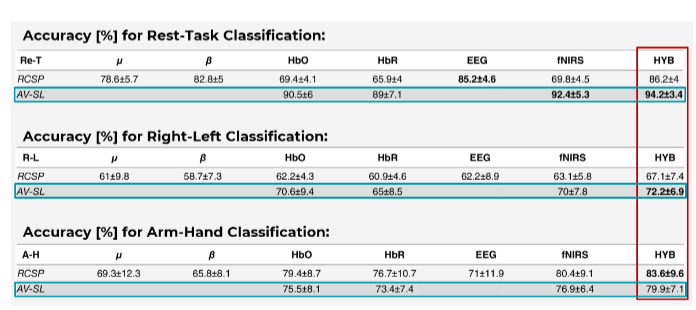
We see a result consistent with that of many other BCI studies: Combining EEG and fNIRS (termed HYB in the table below, highlighted in red) always results in a higher classification accuracy compared to using EEG or fNIRS alone:
Description: “The values are the average accuracy among the 15 subjects ± the standard deviation. RCSP stands for Regularized Common Spatial Patterns (applied with the Generic Learning approach), while AV-SL indicates the use of average and slope indicators as fNIRS features.” Source: Adapted from Buccino et al., 2016.
Interestingly, the study also introduces novel fNIRS features called slope indicators, which allow for an earlier distinction of the different movements when compared to traditional fNIRS-BCIs (down to 3 seconds after stimulus onset). Using these novel features in combination with EEG resulted in higher classification accuracy for two out of the three comparisons, suggesting that slope indicators can be a valuable feature for real-time BCI applications using fNIRS.
This study demonstrates that a setup using both modalities, HD-DOT/fNIRS and EEG, can be a more responsive approach for real-time BCI applications. This holds important implications not only for BCI and Neurofeedback research, but also – prospectively – for clinical practise and patient treatment approaches.
Summary
If you are curious to learn more or have questions about the benefits or the technical aspects of a combined EEG-fNIRS setup, please get in touch with us. We are happy to provide guidance on how our fNIRS systems can be best integrated with your EEG system of choice.
Click here to see our Multimodal fNIRS publication list for more examples
Sources:
Cao, J., Bulger, E., Shinn-Cunningham, B., Grover, P. & Kainerstorfer, J. M. (2023).
Diffuse optical tomography spatial prior for EEG source localization in human visual cortex. NeuroImage, 277, 120210.
Buccino, A.P., Keles, H.O. & Omurtag, A. (2016). Hybrid EEG-fNIRS Asynchronous
Brain-Computer Interface for Multiple Motor Tasks. PLOS ONE, 11(1), e0146610.
Click below to learn more on various applications with NIRS:
- fMRI-concurrent fNIRS
- TMS-concurrent fNIRS
- Customized Real-time Analysis in NIRS: BCI / Neurofeedback
- Combining eye-tracking with fNIRS
- Using NIRS to conduct child and infant measurements